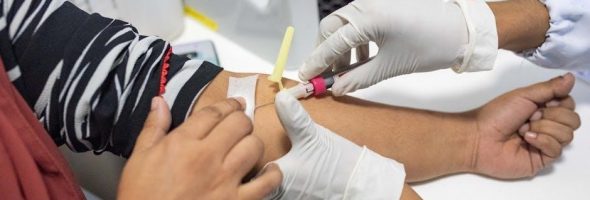
Article Plan: One Blood Therapeutic Phlebotomy Form PDF
This document details a standardized therapeutic phlebotomy form, crucial for consistent blood collection, patient safety, and data integrity –
considering advancements like rusfertide and the “One Blood” initiative.

Therapeutic phlebotomy, a historically significant medical practice, has evolved from ancient bloodletting to a precisely controlled treatment modality. Initially employed for humoral balance, its modern application centers on managing specific hematological disorders. The process involves deliberate blood removal to alleviate disease symptoms and improve patient outcomes.
Contemporary therapeutic phlebotomy isn’t merely a technique; it’s a carefully orchestrated intervention requiring skilled phlebotomy teams and standardized protocols. The “One Blood” initiative emphasizes the importance of consistent procedures and comprehensive documentation, exemplified by the therapeutic phlebotomy form. This form serves as a critical tool for recording patient history, monitoring treatment response, and ensuring patient safety throughout the process.
Understanding the historical context and modern applications is vital, especially considering emerging alternatives like rusfertide, which aim to reduce reliance on frequent phlebotomy. The form’s design reflects these advancements, prioritizing accurate data collection and adherence to ethical guidelines.
What is Therapeutic Phlebotomy?
Therapeutic phlebotomy is a medical procedure involving the controlled removal of blood from the body for therapeutic purposes. Unlike routine blood draws for diagnostic testing, it’s a treatment itself, aimed at reducing excess red blood cells or iron accumulation. This targeted blood evacuation addresses underlying conditions like polycythemia vera and hereditary hemochromatosis, improving patient health.
The procedure requires a trained phlebotomy team, meticulous technique, and careful monitoring. A standardized therapeutic phlebotomy form is essential for documenting patient details, medical history, and the specifics of each phlebotomy session. This form ensures consistency and facilitates accurate tracking of treatment response.
Effective therapeutic phlebotomy minimizes complications through proper patient identification and adherence to safety protocols. The form aids in this process, serving as a vital component of a comprehensive and standardized approach to patient care.
Historical Context of Phlebotomy
Phlebotomy, the practice of bloodletting, boasts a long and complex history, evolving from ancient humoral theory to modern therapeutic applications. Historically, it was a cornerstone of medical practice, believed to restore balance by removing “bad blood.” While often misguided by outdated beliefs, these early practices laid the groundwork for understanding blood’s role in health.
Over centuries, phlebotomy transitioned from a generalized remedy to a more targeted therapy. The recognition of conditions like hereditary hemochromatosis, where iron overload necessitates blood removal, marked a turning point. This shift demanded more precise techniques and documentation – precursors to the modern therapeutic phlebotomy form.
Today, standardized forms ensure consistent practice, patient safety, and accurate record-keeping, a far cry from the often-unregulated practices of the past. The evolution reflects a commitment to evidence-based medicine and optimized patient outcomes.
Modern Applications of Therapeutic Phlebotomy
Therapeutic phlebotomy today is a highly specialized procedure, far removed from its historical roots. It’s primarily employed in managing specific hematological disorders, focusing on precise blood volume reduction to alleviate symptoms and prevent complications. Conditions like polycythemia vera, where the body overproduces red blood cells, are key targets.
The procedure isn’t limited to these core applications; it also plays a role in managing hereditary hemochromatosis, reducing iron overload, and even in certain porphyria cases. Careful monitoring of patient response is crucial, often alongside other therapies like iron chelation or, increasingly, alternatives like rusfertide.
The One Blood therapeutic phlebotomy form is integral to this modern practice, ensuring standardized data collection and facilitating optimal patient care. Accurate documentation is paramount for tracking treatment efficacy and minimizing risks.
Conditions Treated with Therapeutic Phlebotomy
Therapeutic phlebotomy addresses a range of conditions characterized by blood abnormalities. Hereditary hemochromatosis, a common genetic disorder causing iron overload, is a primary indication, with phlebotomy serving as a foundational therapy to reduce iron accumulation and prevent organ damage. Without intervention, this can lead to severe complications.
Polycythemia vera, marked by excessive red blood cell production, also benefits significantly from regular phlebotomy to manage blood viscosity and reduce the risk of thrombotic events. Porphyria cutanea tarda, a rarer metabolic disorder, can be alleviated through controlled blood removal.
The One Blood therapeutic phlebotomy form is vital for meticulously documenting the specifics of each case, including the underlying condition, treatment frequency, and patient response, ensuring consistent and effective care.
5.1 Hereditary Hemochromatosis
Hereditary hemochromatosis, the most prevalent genetic disorder, necessitates careful monitoring and management via therapeutic phlebotomy. This condition causes excessive iron absorption, leading to iron overload and potential damage to organs like the liver, heart, and pancreas. The One Blood therapeutic phlebotomy form is crucial for tracking iron levels and phlebotomy frequency.
Without consistent therapeutic intervention, untreated patients face a significantly reduced median survival rate, particularly those with severe heart failure. Phlebotomy effectively removes excess iron, mitigating disease progression. The form ensures detailed documentation of each session, including volume removed and patient tolerance.
Standardization through the “One Blood” initiative aims to streamline data collection and improve patient outcomes in hemochromatosis management.
5.2 Polycythemia Vera
Polycythemia Vera (PV), a myeloproliferative neoplasm, results in an overproduction of red blood cells, increasing blood viscosity and raising the risk of thrombotic events. Therapeutic phlebotomy serves as a primary treatment, reducing hematocrit levels and alleviating symptoms. The One Blood therapeutic phlebotomy form is vital for meticulously documenting hematocrit reductions and monitoring treatment response.
Recent research highlights rusfertide as a potential alternative, maintaining hematocrit control and reducing phlebotomy dependence over 52 weeks. However, phlebotomy remains a cornerstone of PV management, and the standardized form ensures consistent data collection for comparative analysis.
The form’s detailed record-keeping supports informed clinical decisions and facilitates tracking of long-term patient outcomes in PV treatment protocols.
5.3 Porphyria Cutanea Tarda
Porphyria Cutanea Tarda (PCT), a metabolic disorder, leads to the accumulation of porphyrins in the skin, causing photosensitivity and blistering. Therapeutic phlebotomy effectively reduces iron overload, a key factor in PCT pathogenesis, by removing iron and stimulating erythropoiesis. The One Blood therapeutic phlebotomy form is crucial for tracking iron levels and monitoring the patient’s response to treatment.
Consistent documentation within the form allows clinicians to assess the effectiveness of phlebotomy in reducing porphyrin levels and improving cutaneous symptoms. Careful monitoring is essential, as inadequate iron reduction can lead to disease progression.
The standardized form ensures comprehensive data collection, supporting research and optimizing PCT management protocols.
The Role of Blood Sample Collection
Blood sample collection is central to therapeutic phlebotomy, serving both diagnostic and therapeutic purposes. Accurate and consistent collection, documented meticulously on the One Blood therapeutic phlebotomy form, is paramount. Samples are drawn by a trained phlebotomy team to ensure quality and minimize complications.
These samples facilitate monitoring iron levels in conditions like hemochromatosis and assessing hematocrit in polycythemia vera. Proper blood evacuation techniques are vital, alongside adherence to strict patient identification and safety protocols.
The form’s detailed record-keeping supports effective treatment adjustments and contributes to a comprehensive understanding of the patient’s response to phlebotomy.
Phlebotomy Team Training and Qualifications
Comprehensive training for the phlebotomy team is non-negotiable when utilizing the One Blood therapeutic phlebotomy form. Personnel must demonstrate proficiency in blood evacuation techniques, patient identification, and recognizing potential complications; Training should cover proper use of blood collection tubes and understanding their additives – like lithium heparin – and appropriate inversions.
Qualifications extend beyond technical skills; a thorough understanding of the conditions treated – hereditary hemochromatosis, porphyria cutanea tarda, polycythemia vera – is essential.
The team must be adept at documenting observations accurately on the form, contributing to informed clinical decisions and ensuring patient safety throughout the therapeutic process.
Blood Collection Tubes and Additives
Accurate blood sample collection, documented on the One Blood therapeutic phlebotomy form, relies heavily on selecting the correct blood collection tubes and understanding their additives. Lithium heparin tubes are frequently utilized, preventing clot formation and preserving sample integrity for specific analyses.
Furthermore, tubes designated for Blood Group & Rh testing are crucial for patient safety, especially when repeated phlebotomy is required. Proper inversions of these tubes, as per laboratory protocols, are vital to ensure additive-to-blood ratios are correct.
The form should clearly indicate the tube types used, ensuring traceability and minimizing errors in downstream laboratory processing. Consistent adherence to these standards is paramount.
8.1 Lithium Heparin Tubes
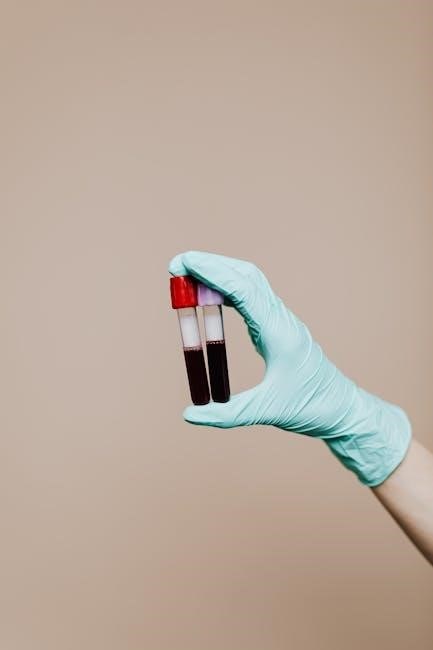
Lithium heparin tubes are a cornerstone of therapeutic phlebotomy, frequently specified on the One Blood therapeutic phlebotomy form. Heparin acts as an anticoagulant, preventing blood coagulation and preserving cellular components for accurate laboratory analysis. This is particularly important when monitoring iron levels in conditions like hereditary hemochromatosis, where repeated sampling is necessary.
The form must detail the correct inversion count – typically 8-10 gentle inversions – to ensure adequate mixing of the heparin with the collected blood. Insufficient mixing can lead to inaccurate results and necessitate recollection.
Proper tube selection and handling, as documented on the form, are critical for reliable diagnostic data and effective patient management.
8.2 Blood Group & Rh Testing Tubes
Blood Group & Rh testing tubes are essential components detailed within the One Blood therapeutic phlebotomy form, ensuring accurate patient identification and compatibility assessments. While not directly related to the therapeutic reduction of iron or blood volume, confirming blood type is vital, especially in cases requiring potential transfusion during or after phlebotomy procedures.
The form should clearly indicate the necessity for proper labeling with two unique patient identifiers, minimizing the risk of inappropriate patient identification. Accurate Rh factor determination is crucial, preventing adverse reactions should transfusion become necessary;
These tubes contribute to a comprehensive safety profile, documented on the form, safeguarding patients undergoing therapeutic phlebotomy.
Blood Evacuation Techniques
Blood evacuation is a critical aspect documented within the One Blood therapeutic phlebotomy form PDF, directly impacting sample quality and patient comfort. The form emphasizes adherence to standardized techniques to prevent hemolysis or inaccurate results. Proper vein selection, tourniquet application, and tube order of draw are paramount, minimizing complications.
The form should detail the importance of a smooth, controlled draw, avoiding excessive force or prolonged tourniquet time. Training and qualifications of the phlebotomy team are vital, ensuring competency in these techniques.
Addressing blood evacuation concerns is central to the form’s purpose, promoting best practices and reducing the risk of incorrect blood collection techniques, ultimately enhancing patient safety and data reliability.
Potential Complications of Phlebotomy
The One Blood therapeutic phlebotomy form PDF necessitates a dedicated section outlining potential complications, ensuring proactive risk management. Common issues include hematoma formation, nerve injury, infection, and patient anxiety. The form should detail procedures for immediate response to these events, prioritizing patient well-being.
Furthermore, the form must address more severe, though less frequent, complications like cardiopulmonary arrest, particularly relevant in therapeutic phlebotomy for conditions like polycythemia vera.
Documentation of patient monitoring during and after the procedure is crucial, alongside clear protocols for addressing inappropriate patient identification or incorrect blood collection techniques that could exacerbate risks. A comprehensive approach to complication awareness is central to the form’s design.
Patient Identification and Safety Protocols
The One Blood therapeutic phlebotomy form PDF must prioritize robust patient identification protocols. This includes requiring at least two unique identifiers – name and date of birth – and verifying against official documentation. A dedicated section should detail procedures for confirming patient consent and explaining the phlebotomy process.
Safety protocols are paramount, encompassing infection control measures like hand hygiene and sterile technique. The form should mandate a pre-phlebotomy assessment of the patient’s medical history, focusing on bleeding disorders or medication use.
Addressing inappropriate patient identification risks is critical; the form should outline steps for resolving discrepancies immediately. Clear documentation of all safety checks performed is essential for maintaining a secure and reliable process.
Inappropriate Patient Identification Risks
The One Blood therapeutic phlebotomy form PDF must explicitly address the severe consequences of inappropriate patient identification. Misidentification can lead to transfusion errors, incorrect therapeutic phlebotomy procedures, and potentially life-threatening complications. The form should detail a multi-step verification process, going beyond simply asking for a name.
Risks include similar patient names, incorrect date of birth information, and reliance on outdated identification. The form needs a dedicated space to document verification steps and any discrepancies encountered.
Protocols for resolving identification uncertainties – such as contacting a physician or reviewing medical records – must be clearly outlined. A checklist ensuring all identification steps are completed before proceeding with phlebotomy is crucial for minimizing errors and ensuring patient safety;
Incorrect Blood Collection Techniques
The One Blood therapeutic phlebotomy form PDF must emphasize the importance of standardized blood evacuation techniques to mitigate risks associated with incorrect collection. Improper technique can lead to hemolysis, inaccurate lab results, and patient discomfort. The form should detail proper venipuncture site selection, angle of insertion, and appropriate tube order of draw.
Insufficient training or failure to adhere to established protocols are key concerns. The form should include a section for phlebotomists to confirm competency and acknowledge adherence to best practices.
Specific attention should be given to avoiding excessive tourniquet time and proper mixing of blood with additives in collection tubes (like lithium heparin). A checklist verifying technique adherence is vital for quality control and patient safety.
Therapeutic Phlebotomy and Chemotherapy
The One Blood therapeutic phlebotomy form PDF needs a dedicated section addressing interactions between therapeutic phlebotomy and chemotherapy treatments. Certain chemotherapeutic agents can impact blood counts, potentially altering the necessity or schedule of phlebotomy. Careful coordination between oncology and phlebotomy teams is paramount.
The form should include fields to document current chemotherapy regimen, recent dosages, and any observed hematological side effects. This information is crucial for determining safe blood removal volumes and minimizing patient risk.
Consideration must be given to potential complications like increased susceptibility to infection or bleeding, especially when both therapies are concurrent. The form should prompt assessment of these risks and documentation of preventative measures.
Therapeutic Phlebotomy and Cardiopulmonary Arrest
The One Blood therapeutic phlebotomy form PDF must incorporate protocols for managing situations involving cardiopulmonary arrest during or immediately following phlebotomy. Documentation should clarify whether arrest occurred during therapeutic blood removal or independently.
A dedicated section should detail immediate cessation of phlebotomy upon arrest, alongside prompt initiation of resuscitation procedures. The form needs fields to record the volume of blood already removed, and any observed signs of bleeding.
Crucially, the form should guide documentation regarding whether bleeding continued post-arrest, and if so, the estimated amount. This information is vital for assessing contributing factors and optimizing future patient care. Clear guidelines for notifying relevant medical personnel are also essential;
Monitoring Patient Response to Phlebotomy
The One Blood therapeutic phlebotomy form PDF requires a comprehensive section dedicated to monitoring patient response. This includes vital signs – blood pressure, heart rate, and respiratory rate – recorded before, during, and after each phlebotomy session.
Specific fields should track symptoms like dizziness, fatigue, or chest pain, allowing for early detection of adverse reactions. Regular hematological assessments, including hemoglobin and hematocrit levels, are crucial, and designated spaces for these results are necessary.
The form must also facilitate documentation of iron studies, particularly in cases of hereditary hemochromatosis, to evaluate treatment efficacy. A space for noting any adjustments to the phlebotomy schedule based on patient response is vital for personalized care.
Rusfertide as an Alternative to Phlebotomy
The One Blood therapeutic phlebotomy form PDF should incorporate a section addressing alternative therapies like rusfertide, a novel treatment for polycythemia vera (PV). This section isn’t to replace phlebotomy documentation, but to record consideration of, or transition to, this alternative.
Fields should document whether rusfertide was discussed with the patient, including benefits and risks, and the patient’s preference. If rusfertide is initiated, the form needs space to track dosage, administration dates, and any observed side effects.
Crucially, the form must allow for continued monitoring of hematocrit levels even with rusfertide use, noting any reduction in phlebotomy requirements. This ensures a comprehensive record of the patient’s overall management strategy.
The “One Blood” Initiative & Standardization
The One Blood therapeutic phlebotomy form PDF is ideally aligned with broader standardization efforts within healthcare, like the “One Blood” initiative. This initiative aims to improve blood management practices and data consistency across institutions.
The form should utilize standardized terminology for conditions treated (hemochromatosis, polycythemia vera, porphyria cutanea tarda), blood collection techniques, and potential complications. This facilitates data sharing and research.
Incorporating unique patient identifiers and adhering to strict data security protocols are paramount, ensuring patient privacy while enabling interoperability. The form’s design should promote accurate and complete documentation, supporting quality improvement and reducing errors in therapeutic phlebotomy procedures.
Therapeutic Phlebotomy Form: Key Components
A comprehensive One Blood therapeutic phlebotomy form PDF must include critical sections for accurate documentation. Patient Demographics (name, DOB, contact info) are foundational, alongside a detailed Medical History section, outlining relevant conditions like hereditary hemochromatosis or polycythemia vera.
The form should document the indication for phlebotomy, volume of blood removed, date/time of procedure, and the phlebotomist’s credentials. A section for documenting patient consent and any observed complications is essential.
Furthermore, recording blood collection tube types (lithium heparin, blood group & Rh testing) and any adverse events ensures a complete patient record. Standardized fields and clear instructions enhance usability and data integrity.
19.1 Patient Demographics Section
The Patient Demographics section of the One Blood therapeutic phlebotomy form PDF is paramount for accurate patient identification and record linkage. This section must include the patient’s full legal name, date of birth, complete address, and reliable contact information – phone number and email if available.
A unique patient identifier, such as a medical record number, is crucial to prevent misidentification. Gender and ethnicity data, collected respectfully and optionally, can support research and healthcare equity initiatives.
Emergency contact details are vital for unforeseen events. Accurate demographic data minimizes risks associated with inappropriate patient identification and ensures proper tracking of therapeutic phlebotomy procedures.
19.2 Medical History Section
The Medical History section within the One Blood therapeutic phlebotomy form PDF is critical for assessing patient suitability and managing potential risks. This section requires detailed documentation of pre-existing conditions, particularly those relevant to phlebotomy, such as hereditary hemochromatosis or polycythemia vera.
A comprehensive list of current medications, including dosages, is essential to identify potential drug interactions. Documented history of bleeding disorders, cardiovascular issues, or cardiopulmonary arrest risk factors is vital.
Prior phlebotomy experiences, including any adverse reactions, must be recorded. This section ensures informed consent and allows the phlebotomy team to tailor the procedure for optimal patient safety and therapeutic efficacy.
PDF Format and Accessibility of Forms
Utilizing the PDF format for the One Blood therapeutic phlebotomy form ensures standardized presentation and broad compatibility across healthcare systems. PDFs maintain formatting integrity, crucial for legal and clinical documentation. However, accessibility is paramount.
The form must adhere to accessibility guidelines, such as WCAG, to accommodate users with disabilities. This includes proper tagging for screen readers, alternative text for images, and sufficient color contrast. Fillable form fields should be clearly labeled and navigable via keyboard.
Digital signatures and secure data encryption are essential for maintaining patient privacy and complying with regulations. A well-designed PDF facilitates efficient data collection and streamlines the phlebotomy process.
Data Security and Patient Privacy
Protecting patient data is paramount when utilizing the One Blood therapeutic phlebotomy form. The PDF must incorporate robust security measures to comply with HIPAA and other relevant privacy regulations. This includes encryption of sensitive information both in transit and at rest.
Access controls should be implemented, limiting form access to authorized personnel only. Audit trails are essential for tracking form usage and identifying potential breaches. Data minimization principles should guide form design, collecting only necessary information.
Secure storage and transmission protocols are vital, alongside regular security assessments and updates. Maintaining patient trust requires unwavering commitment to data security and privacy throughout the phlebotomy process.
Legal and Ethical Considerations
The One Blood therapeutic phlebotomy form must adhere to strict legal and ethical guidelines. Informed consent is crucial; patients must understand the procedure, risks, and benefits before signing the form. Documentation must be meticulous, reflecting accurate patient history and treatment details.
Phlebotomy practices must align with established standards of care, avoiding inappropriate patient identification or incorrect blood collection techniques. Legal ramifications can arise from negligence or breaches of patient confidentiality.
Ethical considerations include equitable access to treatment and avoiding bias in patient selection. The form should facilitate responsible data handling and promote transparency in the therapeutic phlebotomy process, ensuring patient well-being and legal compliance.
Future Trends in Therapeutic Phlebotomy
The future of therapeutic phlebotomy is poised for significant evolution, driven by innovations like rusfertide offering alternatives to traditional phlebotomy for conditions like polycythemia vera. The “One Blood” initiative emphasizes standardization, potentially integrating digital forms and electronic health records for seamless data management.
Advancements in hematological research may identify new conditions suitable for therapeutic phlebotomy. Expect increased focus on personalized medicine, tailoring phlebotomy protocols based on individual patient characteristics.
Telemedicine could play a role in remote monitoring of patients undergoing phlebotomy, enhancing safety and accessibility. The One Blood form will likely adapt to incorporate these changes, prioritizing patient-centric care and efficient data collection.